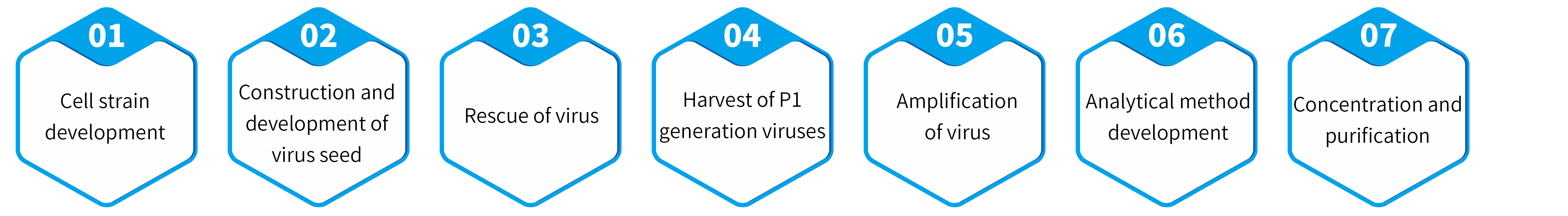
Genevoyager owns the self-developed insect baculovirus
One-Bac 4.0 system independent patent group. One-Bac 4.0 system through
introduction of new strategy of AAV expression element regulation, combined
with key process optimization, achieves high yield, high activity, solid rate, and
single batch capacity up to 1E+18vg in the process of large-scale production of
AAV using insect system.
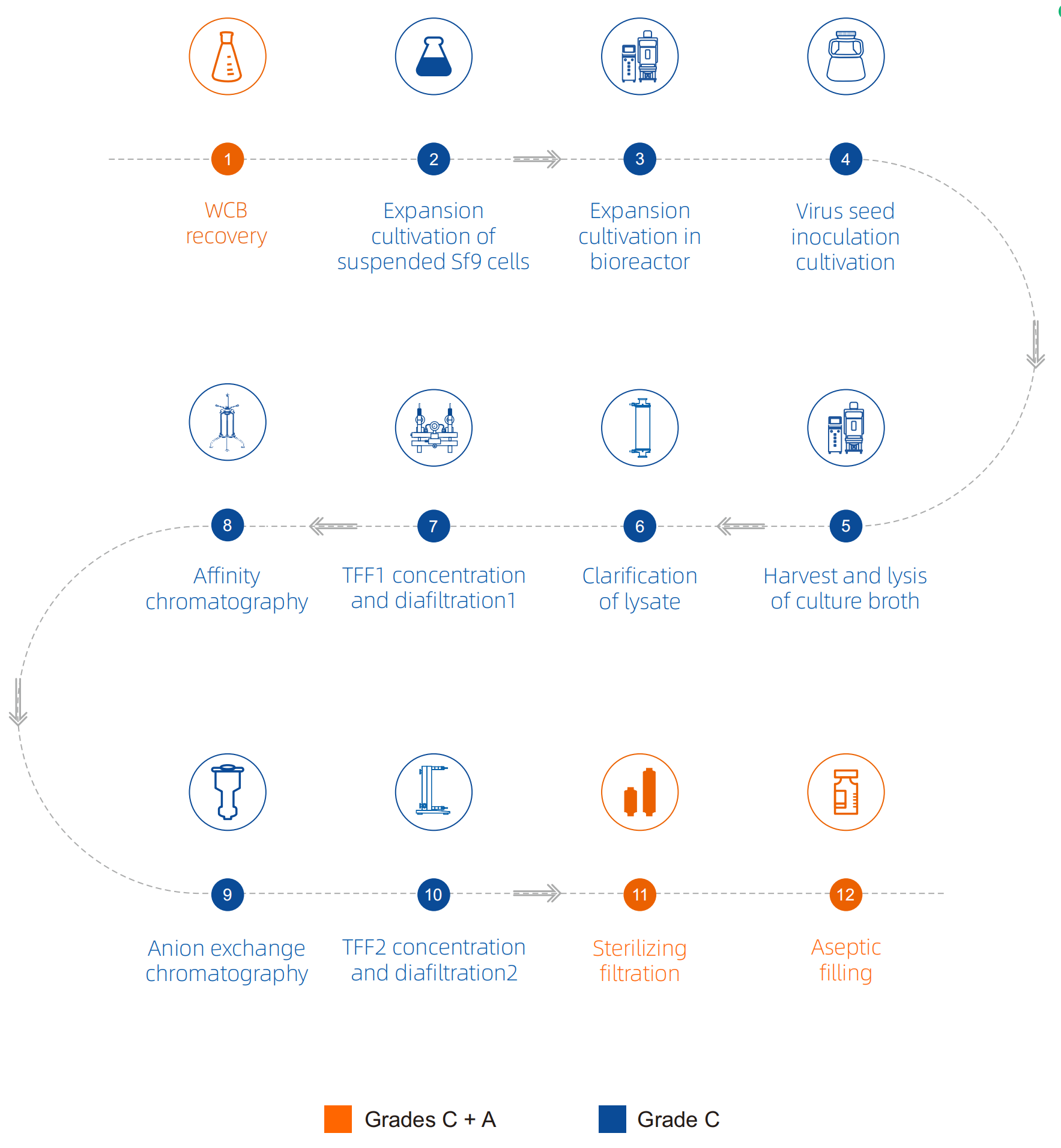
Genevoyager one-stop gene therapy CDMO service platform with professional and experienced QC and QA team can provide perfect quality testing services.
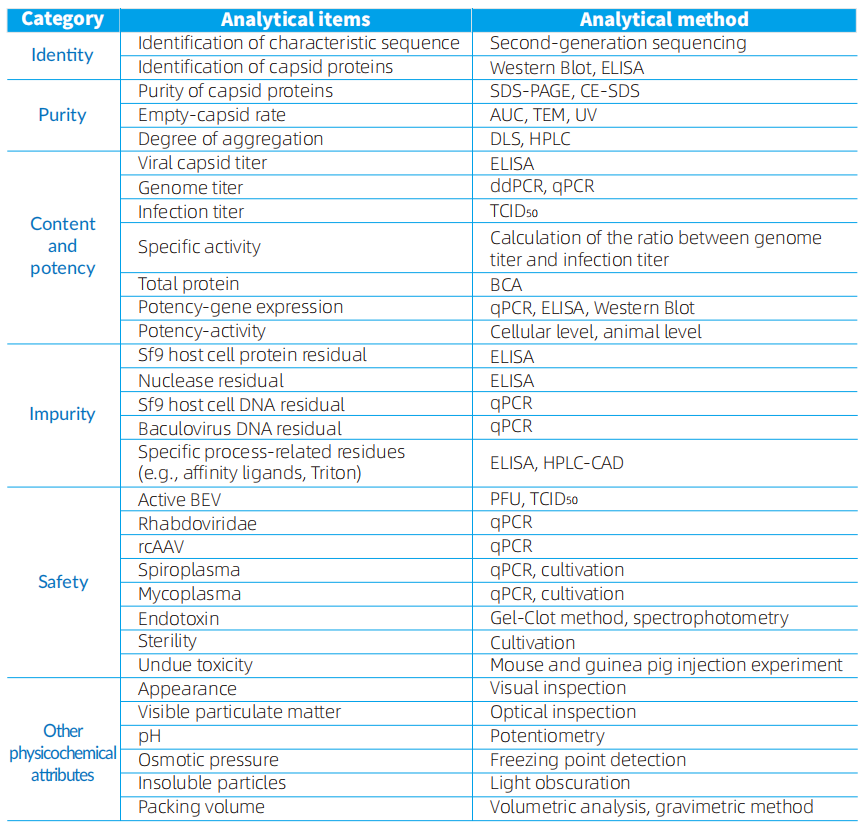
The routine quality release testing items and methods based on AAV are as follows.
The One-Bac 4.0 system produces AAV at an initial yield up to 1~5E+15 vg/L, with stable yields before and after purification at different production scales, and the capacity for a single lot is up to 1E+18 vg.
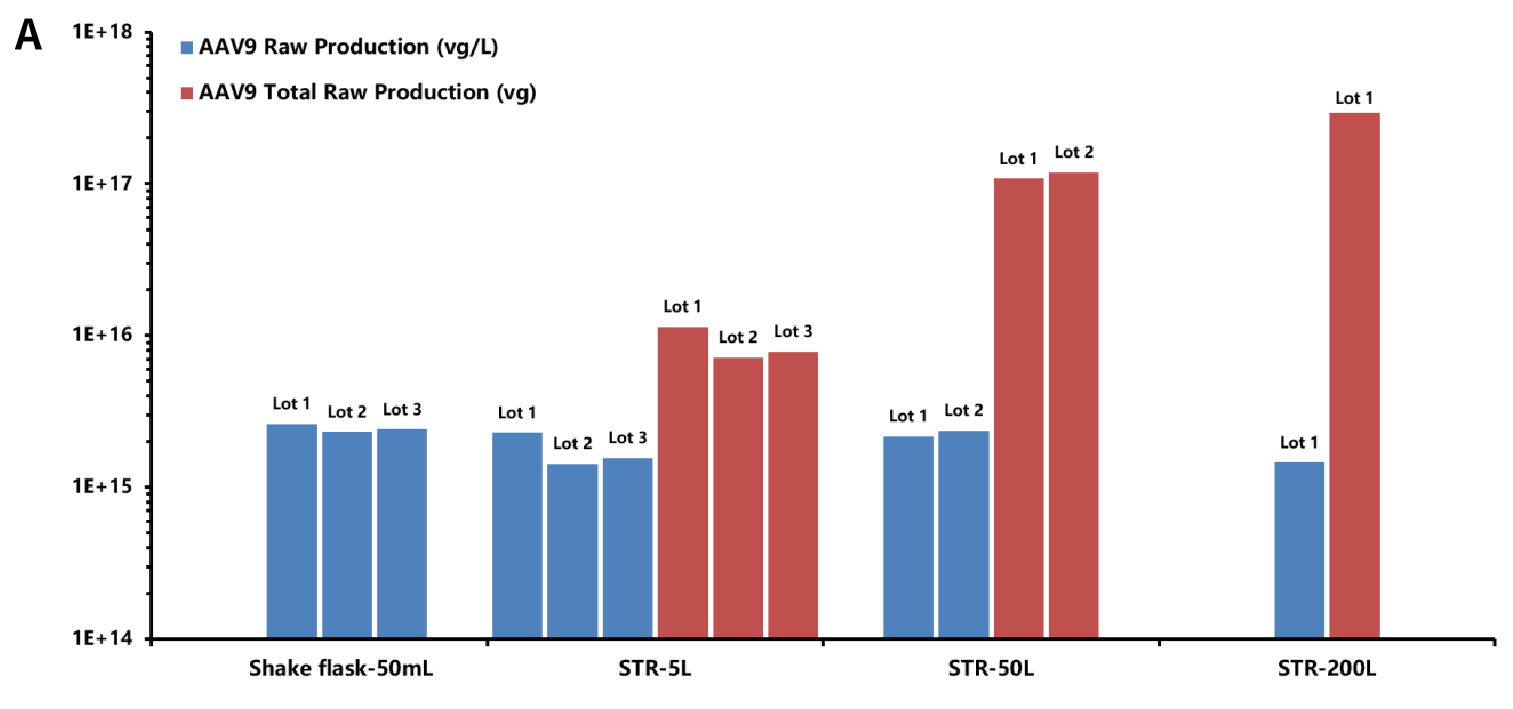
Figure A: Schematic diagram of the initial yield (blue) and total initial yield (red) of AAV from lot to lot at different production scales in a range of 50 mL-200 L (Lot serial numbers represent different lots at the same scale). The initial yield of AAV remains stable at 1~5E+15 vg/L from lot to lot at production scales of 50 mL, 5 L, 50 L and 200 L; the total AAV initial yield is less lossy at linear scaling (from 5 L to 200 L) with good stability from lot to lot.
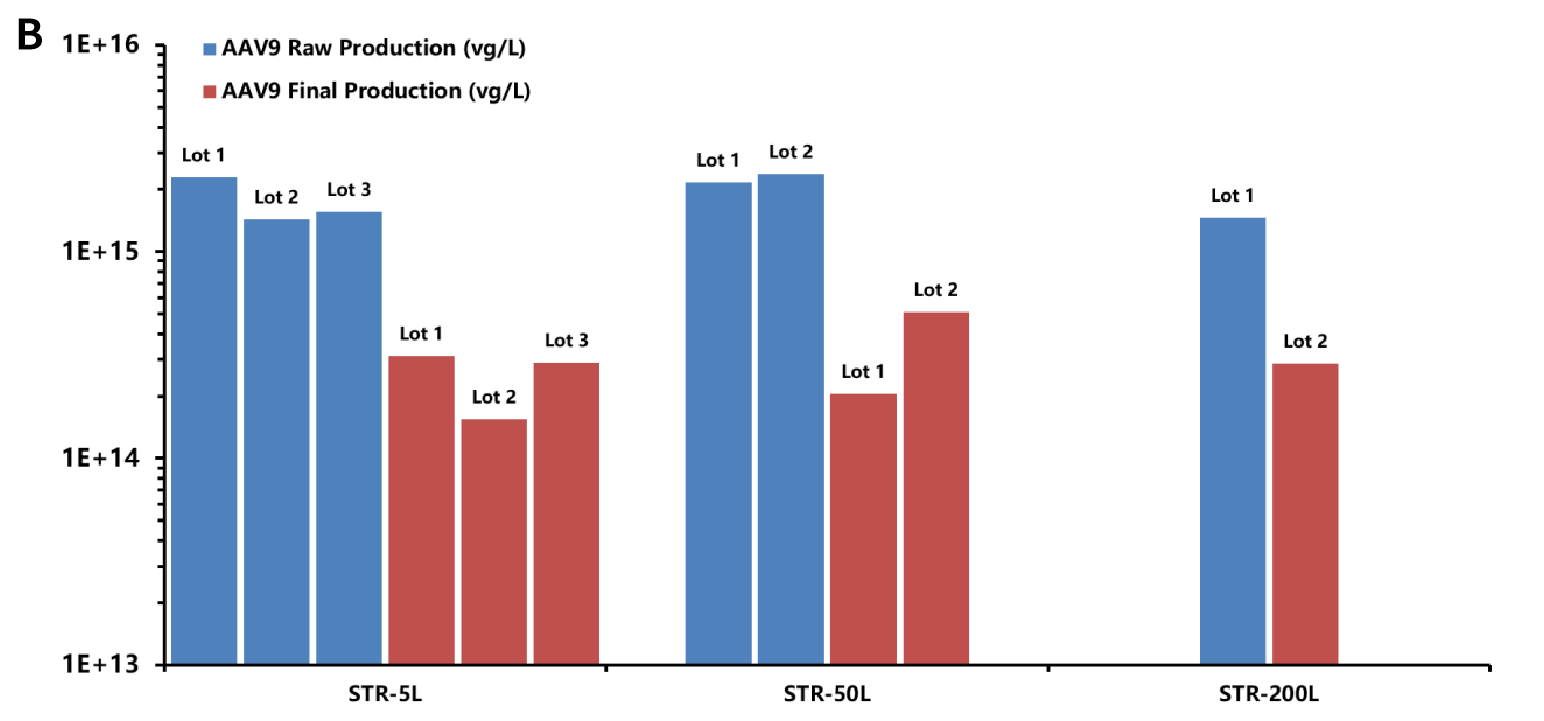
Figure B: Schematic diagram of the initial yield (blue) and post-purification yield (red) of AAV from lot to lot at different production scales in a range of 5 L-200 L (Lot serial numbers represent different lots at the same scale). The post-purification yields of AAV (red) remain stable at 2-4E+14 vg/L at the production scales of 5 L, 50 L and 200 L.
The Baculovirus Expression Vector (BEV) used in One-Bac 4.0 system is highly stable in passage within 10 generations, ensuring that AAV still has a high yield and viral activity from lot to lot when produced on a large scale.
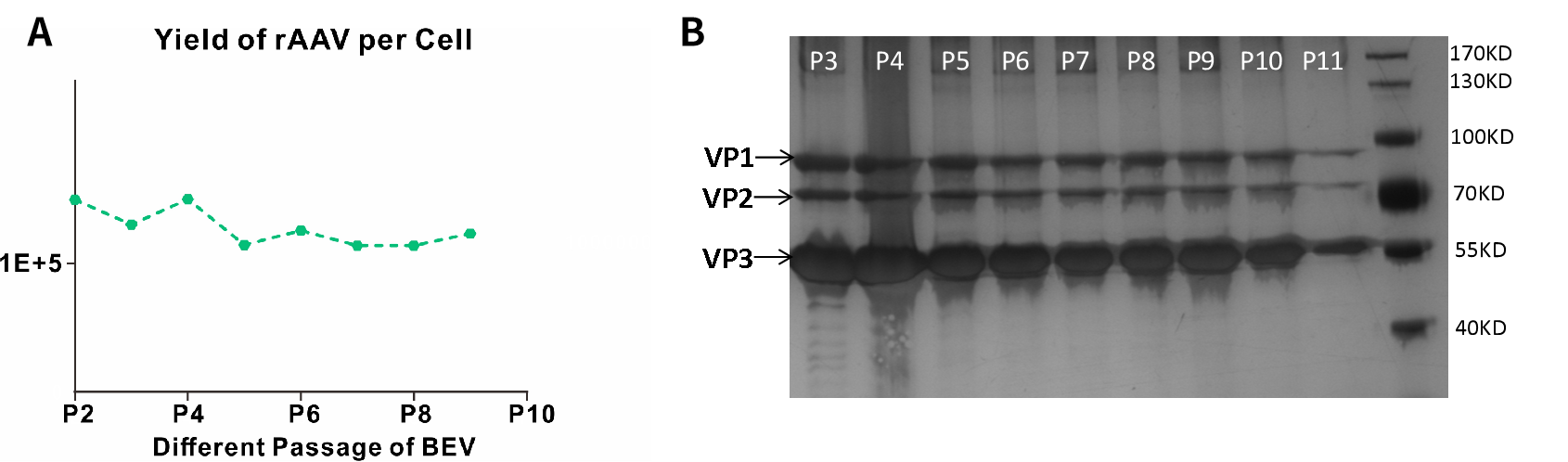
Figure A: Serial passage experiments of BEV showed that a series of passages of BEV for AAV production within P10 generations had no effect on the packaging efficiency of AAV, that is, all BEVs within P10 generations guarantee stable production of more than 1E+5 virus particles from a single SF9 cell, ensuring a stable lot-to-lot yield. Figure B: SDS-PAGE experiments showed that the yield and ratio of viral protein (VP) of AAV expressed by BEV within P10 generations remained stable with VP1:VP2:VP3=1:1:10, ensuring stable virus activity from lot to lot while guaranteeing stable lot-to-lot yield.
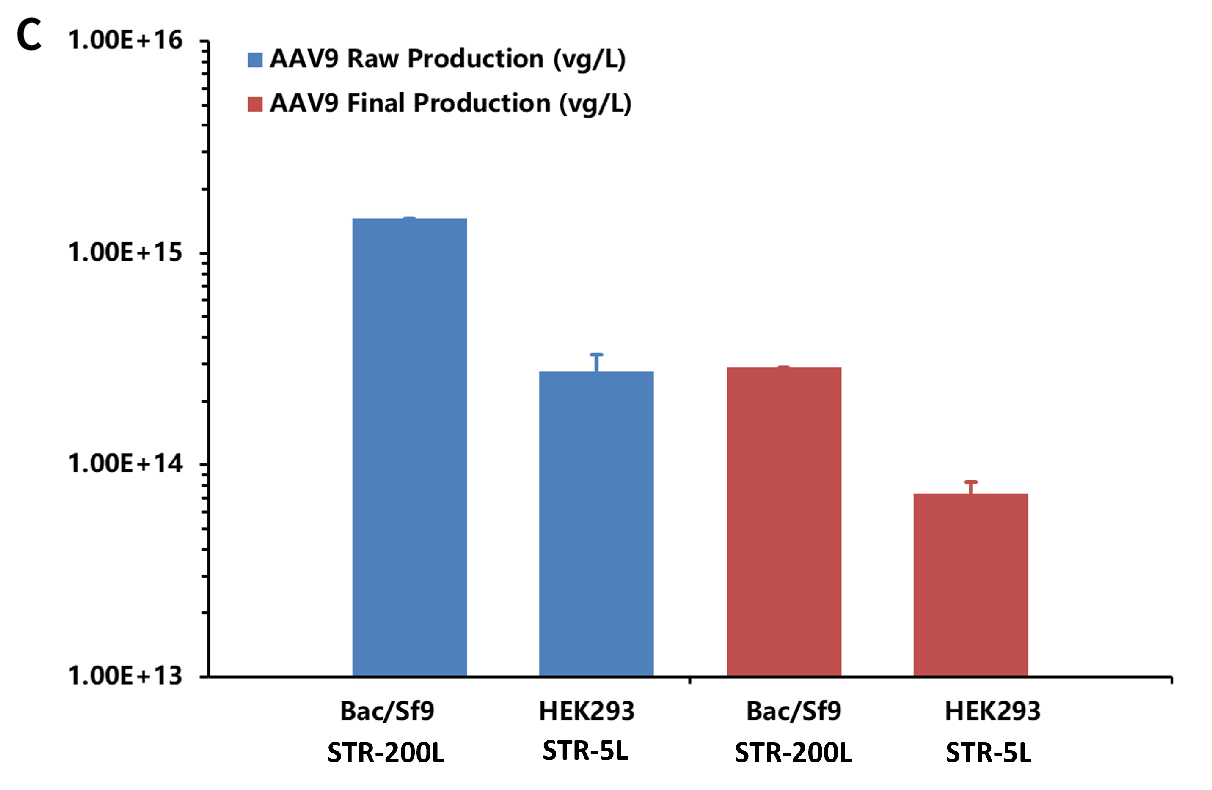
One-Bac 4.0 system has an AAV full-capsid ratio higher than 70% in the harvest fluid and up to 94% in the end product, slightly better than that of HEK293 system.
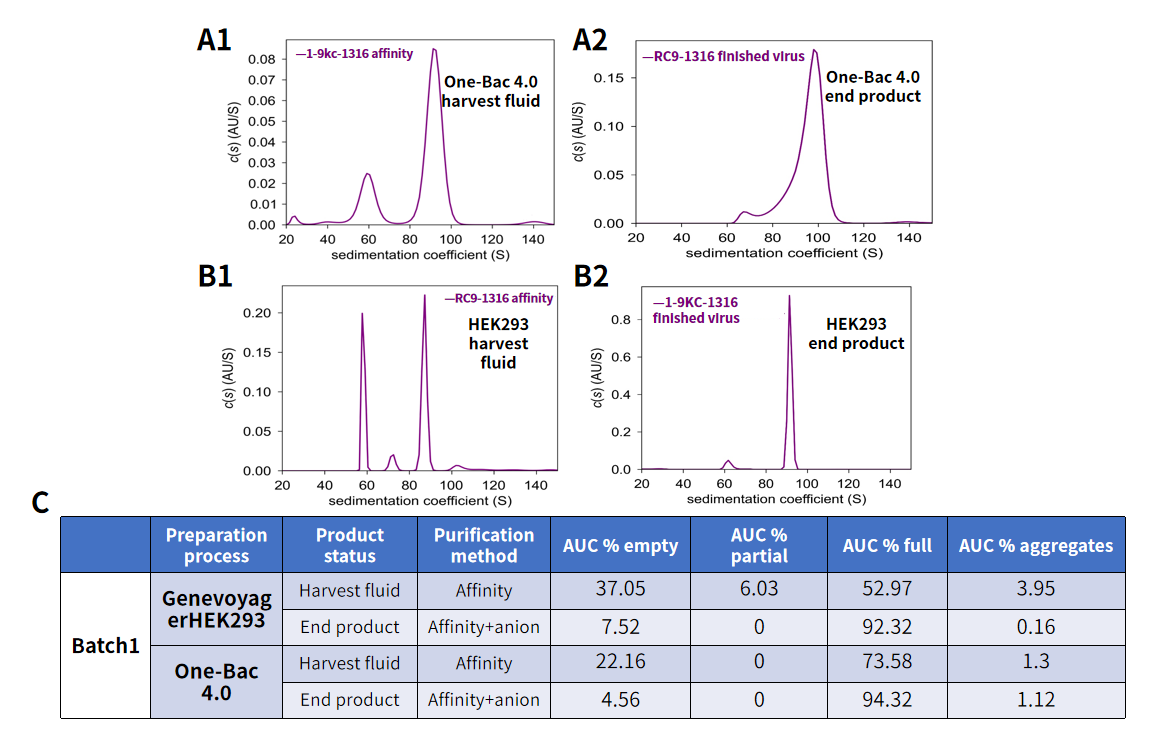
Comparison of AUC full-capsid ratios of harvest fluid and end product AAV between One-Bac 4.0 system and HEK293 system. Figure A: Schematic diagram of AUC results of AAV harvest solution (A1) and end product (A2) for One-Bac 4.0 system. Figure B: Schematic diagram of AUC results of AAV harvest solution (B1) and end product (B2) for Genevoyager HEK293 system. Figure C: Comparison of AUC results of AAV product for One-Bac 4.0 system vs. Genevoyager HEK293 system. In the primary harvest fluid obtained by affinity chromatography, the full-capsid ratio (% full) of AAV reached up to 73.58% for One-Bac 4.0, significantly higher than 52.97% for HEK293 system; in the end product obtained by affinity and anion exchange chromatography, the full-capsid ratio (% full) of AAV reached up to 94.32% for One-Bac 4.0, comparable to 92.32% for HEK293 system. It indicates that AAV produced by One-Bac 4.0 system has a high full-capsid ratio.
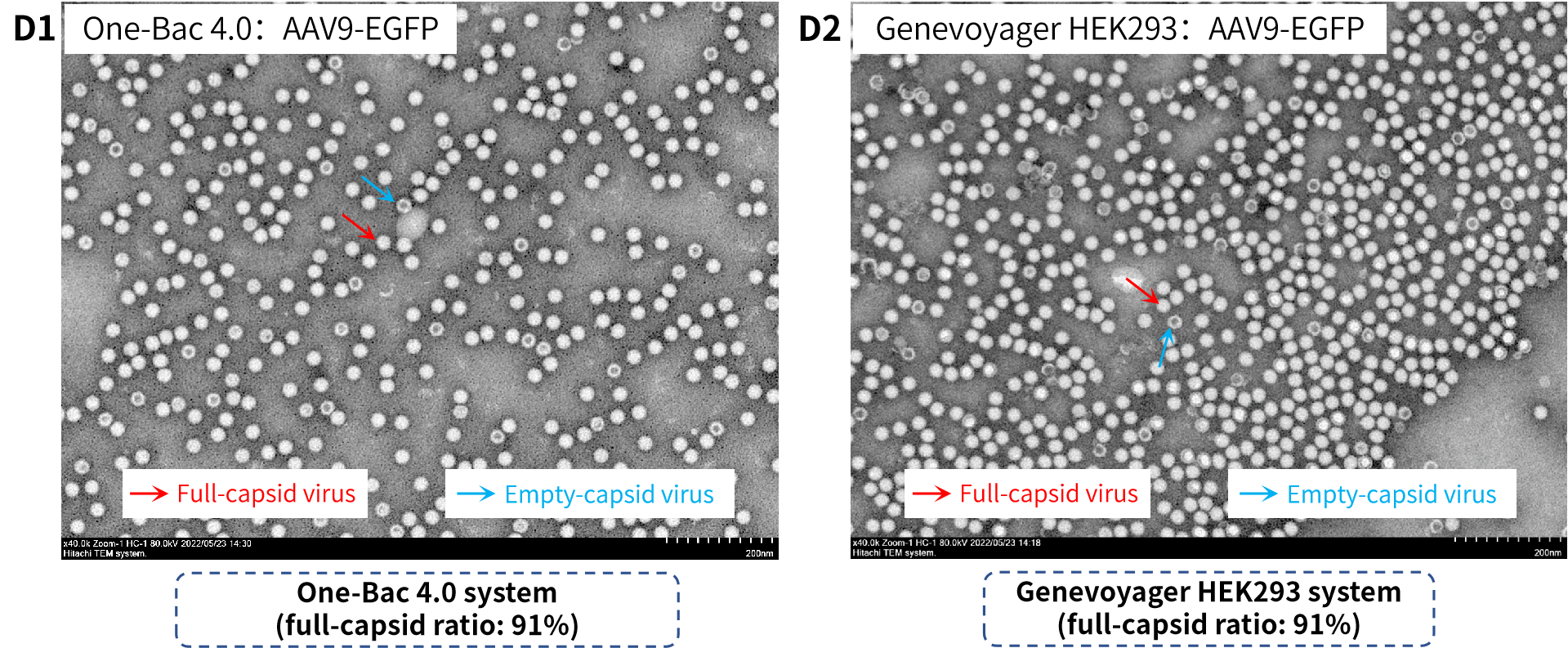
Figure D: Comparison of full-capsid ratios of AAV end products of One-Bac 4.0 system (D1) and Genevoyager HEK293 system (D2) by transmission electron microscopy (TEM). In addition to the AUC test as “a gold standard”, the test results by commonly used TEM also showed that AAV end products packaged by One-Bac 4.0 system had a high full-capsid ratio up to 91%, comparable to that of HEK293 system.
The results of both cell and animal experiments showed that the rAAV infecঞon effffect produced by the One-Bac 4.0TM system was comparable to, or even superior to, that of the HEK293 system.
1. The advantages of high yield and infectious activity of One-Bac 4.0 system make it further reduce the cost of fermentation at scale compared to HEK293 system.
2. One-Bac 4.0 system lowers the raw material cost for mass production of AAV by optimizing the production process without GMP plasmids and high-cost transfection reagents.
The establishment of a virus seed bank has made the virus production process faster and more convenient by simplifying the viral seed infection process. This feature becomes increasingly apparent as the demand for viruses grows, greatly shortening the virus production and delivery timeline.

US: 3675 Market Street, Suite 200, Philadelphia, PA19104 Tel: +1 (215) 205-6963 | +086 027-65023363
E-mail: hui.wang@genevoyager.com
China: No128, Guanggu 7th Rd, East Lake High-tech Development Zone, Wuhan, China Tel: 17720522078
E-mail: marketing@genevoyager.com